Nicole Villalpando Austin American-Statesman – view article
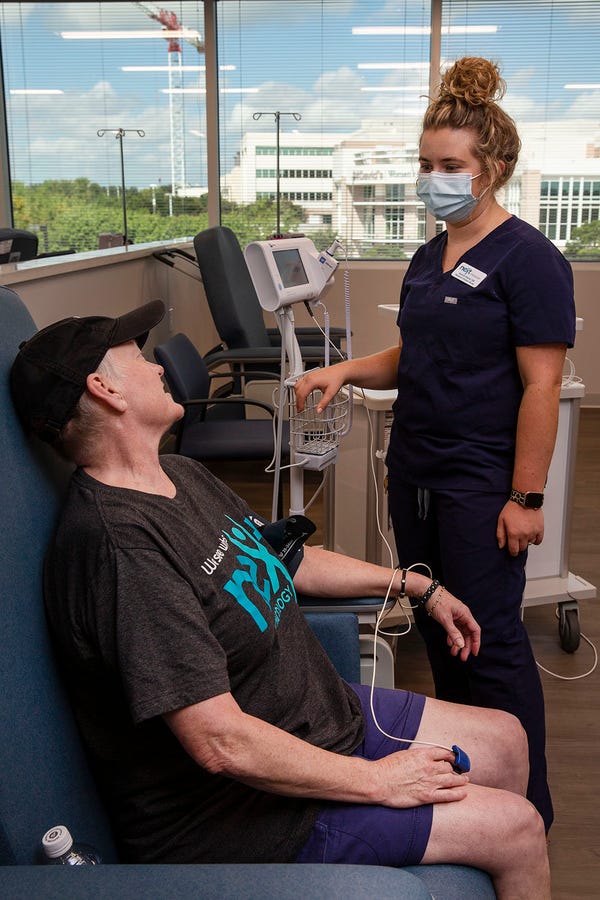
Lynne Xavier, 55, remembers when her husband was part of clinical trials for rectal cancer at MD Anderson in Houston.
“Watching him to go back and forth and back and forth and the wear on him,” she said, made the Austinite decide not to participate in any clinical trials out of town when she was diagnosed with stage 4 lung cancer in 2018.
Xavier’s doctors at Texas Oncology referred her to a new clinic, Next Oncology, that opened officially in July in North Austin. It’s the second location for the clinic, which began in San Antonio and has plans to open in Dallas, Virginia and two sites in Europe in the coming year.
Next Oncology specializes in phase one clinical trials for cancer treatments. Before, people in Austin have had to go to Houston, Dallas or San Antonio or out of state to participate in many of these trials.
“Austin is not the small city it was 20 years ago,” said Dr. Anthony Tolcher, Next Oncology’s founder. “It really hasn’t had a high profile phase one clinic.”
In the past five years, Austin has been experiencing explosive growth in its medical offerings as hospital systems and the University of Texas Dell Medical School’s clinical arm, UT Health Austin, begin offering new programs. These include organ transplant programs, burn treatment programs, and cardiac and stroke care initiatives. Clinical research like the kind at Next Oncology is part of that growth.
Having a clinic that does these kinds of trials in her backyard, “is everything,” Xavier said.
For the past three years she has done multiple treatments, and she had reached a point where doctors didn’t have much to offer her, Xavier said. The one thing that might be an option might be only 10% to 15% effective, she said.
Xavier began participating in a clinical trial three weeks ago, and even though “there’s no way to know” how effective it will be, “the potential is good,” she said.
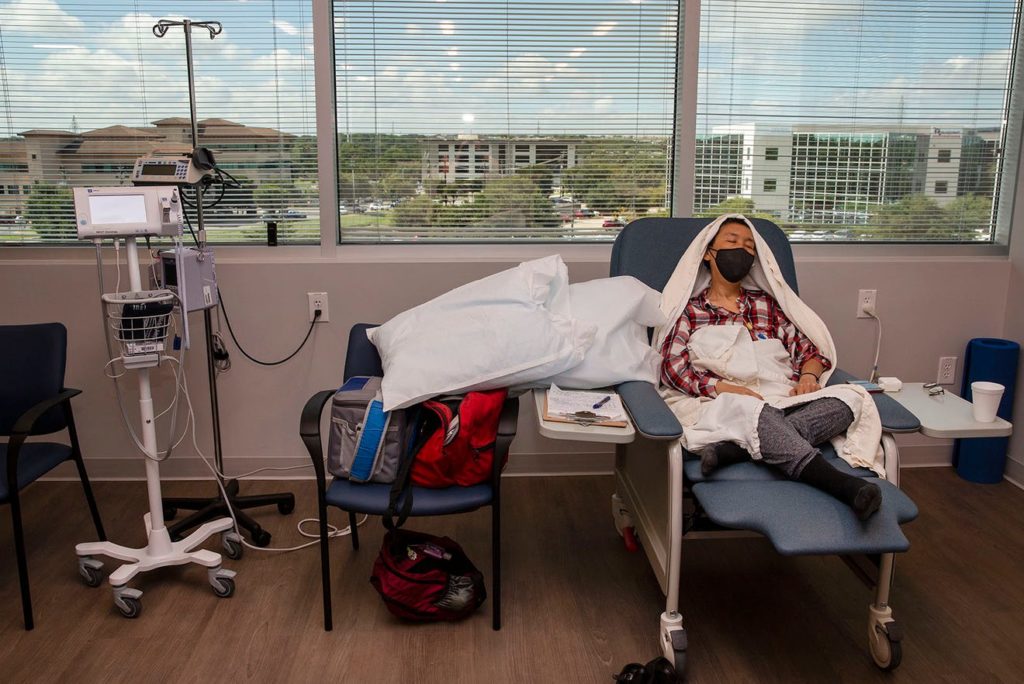
She’s doing a lot of initial lab work now that makes for long days at the clinic and then will get the study drug once a month.
Tolcher and Dr. Andrae Vandross, who is running the Austin Next Oncology site, said they have 16 studies currently.
“This is a portfolio of clinical studies that Austin has never had,” Tolcher said.
Clinics that run phase one trials need to have a specialized pharmacy with the proper machines to mix chemicals as well as highly specialized storage systems and a pharmacist who knows how to maintain those. It also has to have a specialized laboratory and people who specialize in managing the data in compliance with Food and Drug Administration regulations.
“It’s a big undertaking,” Tolcher said. “It requires a big commitment.”
Phase one clinical trials are the initial human studies for new medications, but unlike later phases, no one receives a placebo, said Next Oncology Director of Clinical Operations Marlena Griffin. Researchers are looking for initial dosing recommendations, and they start with low doses and then escalate dosing while looking at side effects and efficacy.
Most drugs that have good results in the phase one trials go through phase two and phase three trials before being approved by the FDA. This process can take 10 to 15 years, Griffin said.

In Austin, Next Oncology staffers hope to enroll 100 people in about 25 phase one trials in the first year and build to 200 patients and 50 trials. The San Antonio site has 200 to 300 patients a year in about 80 trials. The San Antonio site had been seeing about 50 Austin-area patients a year before the Austin clinic opened.
The Austin clinic isn’t just open to Central Texas patients. Kimberly Gibbs, 55, flies down once a week from just north of Tulsa, Okla., to participate in a study for ovarian cancer. She found this study on a database of clinical trials kept by the National Institutes of Health after trying about 30 other treatments in the six years since she was diagnosed.
“I really ran out of options,” Gibbs said.

This study was offered in 10 location in the United States, and Austin was the closest. She could have gone to MD Anderson in Houston, but she said, “It’s just so big.”
Each week is different. Sometimes Gibbs is in the Austin office only one to three hours; sometimes she’s there for 12 hours based on what labs and scans need to be done for the study.
She said she often gets calls from Vandross to check up on her during the week, and she’s become close to the clinical staff since she started the study in July.
“They are nothing but loving and kind,” Gibbs said of the staff.
“I feel very fortunate.”
Study participants get the treatments for free as well as some of the lab work, scans and doctor visits that are considered study-specific. Other treatments that would be part of the standard cancer care are billed through a participant’s insurance.
Vandross stays in close contact with a patient’s oncologist and shares scan and lab results.
Next Oncology also works with local oncologists to find people to participate in studies. Texas Oncology, which has 210 locations throughout the state, is a shareholder in Next Oncology.
Tolcher hopes that as Dell Medical School builds out its oncology program at the LiveStrong Cancer Institutes, it is also able to build out its phase one research program as well as train more researchers and doctors interested in doing these kinds of studies.
“Having more than one phase one center is not a bad thing,” Tolcher said. “We want patients to have more options.”


